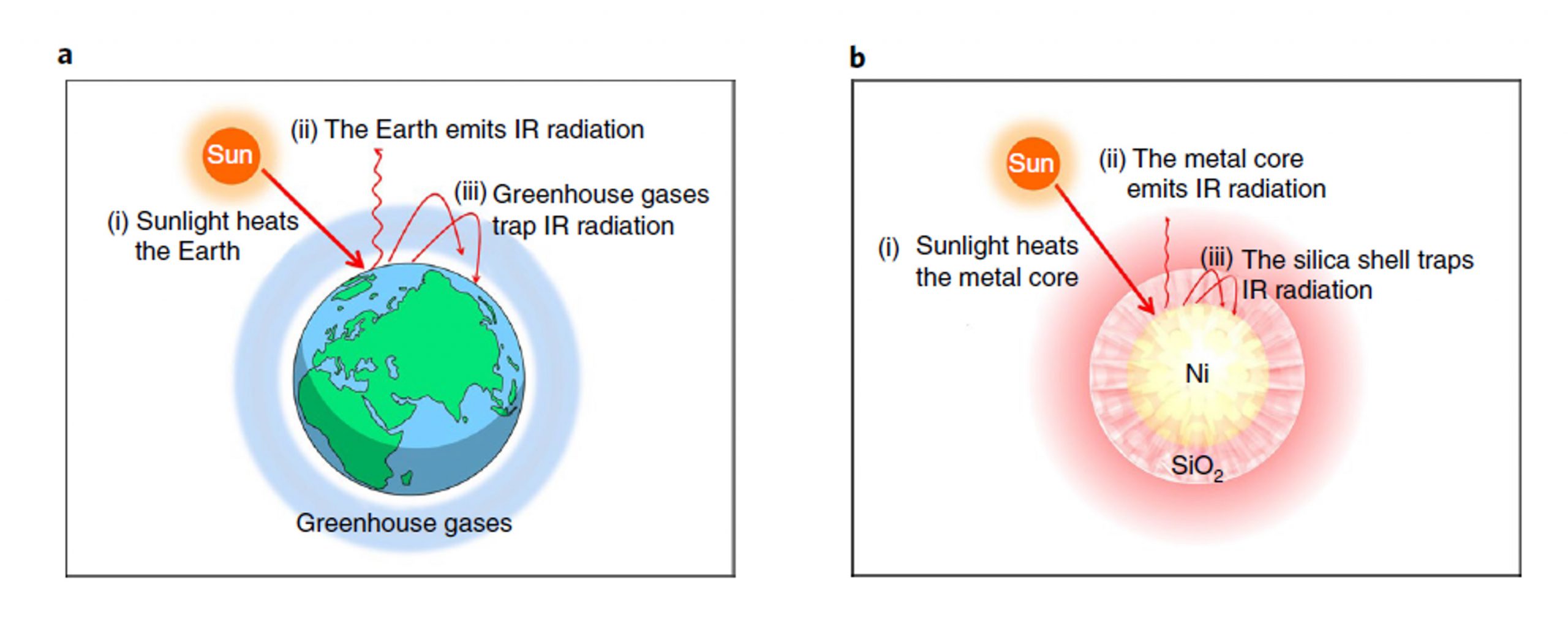
Solar energy and CO2 utilizations are recognized as key to counter the global warming and build a “net-zero” sustainable society, and the surging global temperature has set the deadline of establishing revolutionary techniques in several decades. The urgency requires to assess most feasible current pathways among various solar energy and CO2 proposals and stick to it progressively. Athan Tountas, Prof. Ozin and Prof. Sain recently published their perspective ‘Solar methanol energy storage’ in the Nature Catalysis focus issue ‘CO2 Reimagined’ that acknowledges the five-year anniversary of the Paris Agreement. In it they explore the feasibility of storing renewable energy in the form of intermittent solar energy as methanol and highlight the performance and efficiency advantages of using CO-rich syngas derived from commercial-ready RWGS technology compared to direct-CO2 processes. The Focus is dedicated to progressing the fundamental science and practical implementation of this technology to advance climate goals.
-
Recent Posts
- Congratulations to Geoff’s birthday paper of CO2 photocatalysis on Matter
- Could modified train cars capture carbon from the air? This team has a plan to make it happen
- Sand batteries that are dirt cheap
- Congratulations to Lu, Chengliang and Geoff on their recent publication in Nature Communications
- Congratulations to Wei and co-authors on their recent publication on silica in the Chem Catalysis
Recent Comments
Categories
Header Courtesy of Digital Westex